Pneumology - Case Reports
Vol. 93 No. 3 (2023)
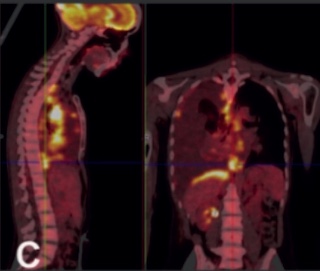
Alectinib rescue therapy in advanced ALK rearranged lung adenocarcinoma: a case report
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Received: 27 July 2022
Accepted: 14 September 2022
Accepted: 14 September 2022
1843
Views
379
Downloads







