Rural treatment of COVID-19 patients with pirfenidone, nitazoxanide and colchicine. Case series
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Accepted: 16 May 2022
Authors
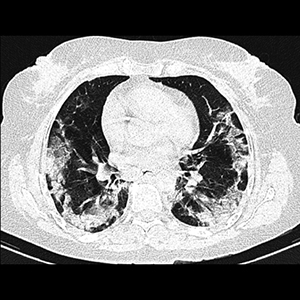
Combined treatments against SARS-CoV-2 are emerging and some have taken into account the post-COVID-19 fibrosis. The aim of this survey was to report the experience of treating COVID-19 patients with pirfenidone, nitazoxanide (NTZ) and colchicine. It was a case series report of COVID-19 patients treated from December 2020 to March 2021, in a rural health center located in the State of Mexico, Mexico. 23 patients were included (mean age 44.5 ± 17.1 years), 12 women (mean age 45.9 ± 17.9 years) and 11 men (mean age 43 ± 16.9 years) with four deaths (17.39%). The evolution time was of 17.3 ± 6.7 days being the main symptoms fever (82.6%), myalgia (69.6%) and cough (65.2%). The main comorbidities were overweight/obesity 18 (78.26%), type 2 diabetes mellitus (T2DM) 4 (17.39%), Chronic obstructive pulmonary disease (COPD) 5 (21.73%) and systemic hypertension 2 (8.69%). Two patients were intubated and both died; in these cases, they refused to take NTZ until after three days the medical doctor had prescribed it for the first time. It can be concluded that implementing a mixed treatment with pirfenidone, NTZ and colchicine could improve the survival rate in ambulatory patients of low socioeconomic status.
How to Cite

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






